

Oxygen is a colorless, odorless and tasteless gas that supports life and combustion. All elements, except rare gases, react with oxygen, over a wide range of temperatures, to form oxides. Oxygen is 1.1 times heavier than air and is slightly soluble in water.
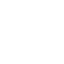

| Units | Weight in Kg | Gas in Nm3 | Liquid in Ltrs |
|---|---|---|---|
| 1 Kg | 1.0 | 0.6996 | 0.8762 |
| 1 Ton | 907.2 | 635 | 794.5 |
| 1 Nm3 Gas | 1.4282 | 1.0 | 1.2511 |
| 1 Ltr Liquid | 1.1416 | 0.7995 | 1.0 |
Bhoruka, is a versatile group & has been on the forefront for over four decades with various activities spread across the country. Mr. P.D. Agarwal (1920-1982) was the founder of the Group & was a great philanthropist and humanitarian. The Bhoruka Group houses the first Company in the private sector to successfully set up a hydro power station in the post-independence era and has also founded companies in various sectors such as Steel (Engineering & Stevedoring), Industrial Gases, I.T. Parks, Education, Community Welfare and allied activities.